The SNP team has just published an article entitled “Insights into the structure and evolution of the human SAGA complex by affinity-ligand purification” in Science Advances.
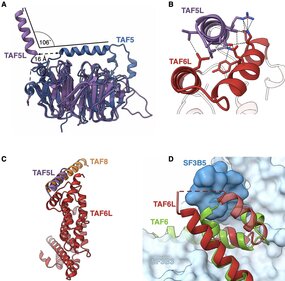
Human SAGA is a 20-subunit transcriptional coactivator. We report the structure of endogenous human SAGA purified via an affinity ligand from cells that were not disturbed by any genomic engineering tools. Our work reveals the high-resolution structure of SPL and the TAF6L HEAT repeat domain that provides the SPL with a docking surface. We elucidate how SPL and the HEAT repeats are incorporated into SAGA. We identify major structural differences between TAF6L/TAF5L and their canonical paralogs that enable SPL accommodation. SPL engages SAGA through a substantially smaller interface than in U2snRNP, despite sharing a deeply inserted helical motif. The seemingly weaker interaction of SPL with SAGA raises the possibility that SAGA relays this module to the splicing machinery.